A complete buyer's guide that provides comprehensive insights on Clinical Trials Investigator Portal License Agreements category spend, spend growth and regional segmentation; in-depth price trends; negotiation levers and analysis of Clinical Trials Investigator Portal License Agreements suppliers.
GET FREE SAMPLEClinical Trials Investigator Portal License Agreements is an essential part of the Science and Environmental Research category which includes all spend related to research of scientific, medical, technological and environmental subjects for commercial or social purposes.
Several macro changes in the dynamics of overall Science and Environmental Research as well as Clinical Trials Investigator Portal License Agreements procurement trends are poised to change the business practices and procurement thumb rules. The growing shortage of skilled talent and increasing salaries of employees is driving the operational costs of service providers. This is expected to increase service charges at least within the tier 1 suppliers who put a high premium on the talent pool that utilize. There are only a handful of \"Tier 1\" vendors in the Clinical Trials Investigator Portal License Agreements market and they have traditionally enjoyed a premium in their pricing. However, Tier 2 suppliers are giving them a tough competition now as they can provide similar services at a lower cost. Many tier 2 suppliers achieve this parity by finding right talent at a relatively lower cost and maintaining lean operations without any frills. Proliferation of supply pool in regions such as APAC is expected to intensify supply market competition. This will compel service providers to compete on price points to attract new buyers, which is expected to offset some of the increase in procurement cost arising out of increasing supplier OPEX. Category managers need to take note of the potential of these developments and reassess the changes required in their procurement practices.
Sometimes, procurement functions are unable to timely alter their practices while responding to market conditions. Industry experts acknowledge that periodically reviewing procurement best practices and adopting learnings from across procurement categories can help procurement teams respond to market needs in a more agile way. This report combines our experience of other categories with Clinical Trials Investigator Portal License Agreements procurement insights and hand picks best practices that can work for category managers delving in this market. For example, Buyers must provide all key requirements in detail and ensure that service providers offer precise details regarding processes, systems, manpower, and expected outcome. Regular and effective communication between buyers and service providers are crucial during the contract formation phase, as well as across different phases of the project. Buyers can optimize the costs by arriving at right sourcing mix through an alignment of in-house capabilities with outsourced functions. Buyers can use in-house capabilities for project-specific requirements that can be resolved internally. Third-party providers should be engaged based on their expertise and project-specific requirements that require capabilities not present in the buyers' organization. Buyers can engage in pilot projects with interested service providers to assess their capabilities. This also helps buyers to assess the cost benefits and value proposition of the proposal.
Activate your free account to gain easy access to cutting edge research and insights on consumers, emerging price trends, global and regional suppliers.
Clinical Trials Investigator Portal License Agreements procurement managers also need to proactively identify and mitigate potential risks that can arise in the supply chain or contracts for Clinical Trials Investigator Portal License Agreements procurement. Some examples include:
For detailed insights and complete access to our report library, activate your free account!
The report is intended to serve as a one-stop reference guide for Clinical Trials Investigator Portal License Agreements procurement strategy and offers a perfect blend of category basics with deep-dive category data and insights. Therefore, it is ideal for category beginners looking for “Clinical Trials Investigator Portal License Agreements: Procurement Report 101” as well as for category experts actively tracking the global Clinical Trials Investigator Portal License Agreements procurement market.
You may have just initiated your research to design a winning Clinical Trials Investigator Portal License Agreements procurement strategy, or you may be a category expert looking for strategic insights and updated data.Either ways, the report has your requirements covered.

Unlock SpendEdge's comprehensive procurement report collection with ease through our procurement platform.
Procurement decisions can prove to be costly in the absence of careful deliberation and evaluation of every available option. In fact, more than 90% of the decision makers we work with acknowledge that timely availability of up-to-date category intelligence can help them make better purchasing decisions. More than 80% of them believe that in-house category intelligence needs to be updated periodically to achieve full benefits. If you have read so far, we are quite sure you agree!!
The Clinical Trials Investigator Portal License Agreements procurement report helps take more informed decisions by placing all the critical information and advice at the fingertips of a decision maker. It also specifically answers some of the key questions that we have been routinely asked during our industry outreach initiatives:
SpendEdge Insights has helped procurement professionals and sourcing teams manage multiple spend areas and achieve more than $2 billion in savings. Activate your free account today!
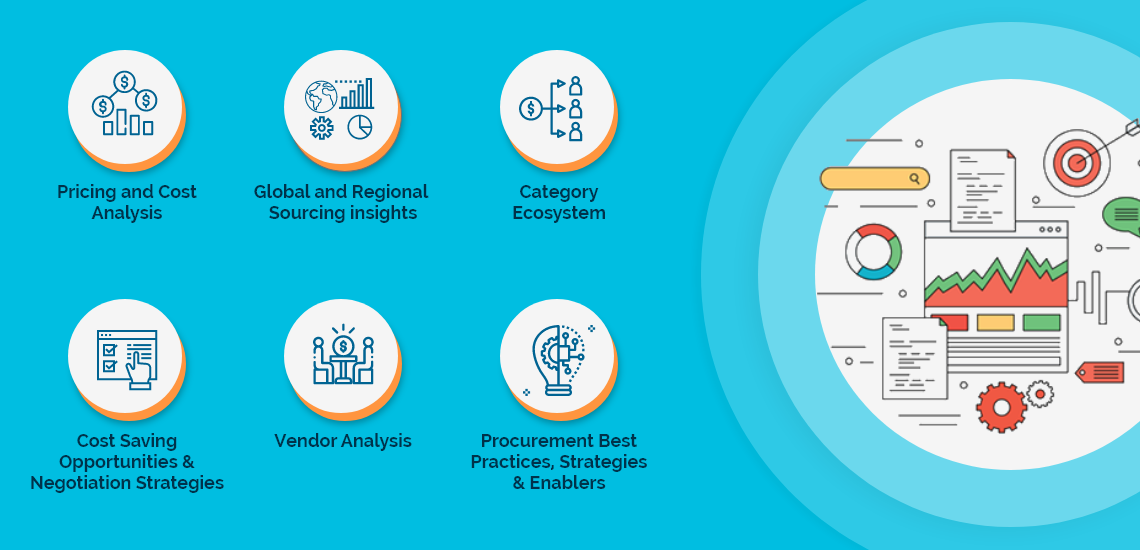
The Clinical Trials Investigator Portal License Agreements market report offers a complete picture of the supply market and analyzes the category from the perspective of both buyers and suppliers. Analysis of the category trends, procurement best practices, negotiation levers and overall category management strategy advisory are interspersed with in-depth data and commentary on spend outlook, pricing ecosystem and supplier landscape drilled down to a region-level coverage.

A key highlight of this report is the in-depth outlook created on Clinical Trials Investigator Portal License Agreements procurement spend and pricing trends. The report further delves deep into the aspects of cost structure, total cost of ownership and supplier margins for Clinical Trials Investigator Portal License Agreements. A dedicated section to supplier profiles and evaluation helps decision makers cast a wider procurement net and identify gaps in existing relationships.

Along with specific category and supplier intelligence, the publication also includes curated insights on Clinical Trials Investigator Portal License Agreements market trends, price influencers and inherent risks. These insights help the decision makers prepare for market shaping trends in advance and create alternative strategies for changes in the market conditions.

Additionally, the report also advises on the best practices and strategies to manage the Clinical Trials Investigator Portal License Agreements category efficiently. Negotiation levers and opportunities are explained in detail along with quantification of their potential. Benchmark KPIs for supplier and buyer performance management are also aggregated to better organize the category objectives. Other themes of advisory include ideal procurement organization structure, enablers to achieve KPIs or category objectives and ideal SLAs to have with suppliers.






Our research is complex, but our reports are easy to digest. Quantitative analysis and exhaustive commentary is placed in an easy to read format that gives you an in-depth knowledge on the category without spending hours to figure out “what does it mean for my company?”
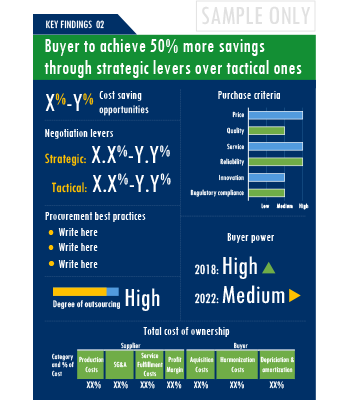
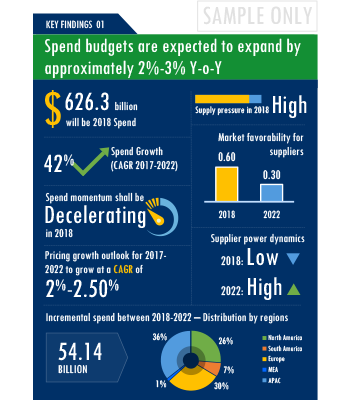
SpendEdge presents a detailed picture of Clinical Trials Investigator Portal License Agreements procurement solutions by way of study, synthesis, and summation of data from multiple sources. The analysts have presented the various facets of the market with a particular focus on identifying the key category influencers. The data thus presented is comprehensive, reliable, and the result of extensive research, both primary and secondary.

Global Outsourced Phase IV Studies Market - Procurement Intelligence Report

Global Phase I / Clinical Pharmacology Unit Services Market - Procurement Intelligence Report

Global Clinical Supply Management Market - Procurement Intelligence Report

Global Research Preformulation Services Market - Procurement Intelligence Report

Global Interactive Response Systems (IxRS) Market - Procurement Intelligence Report

Global Clinical Trials Investigator Portal Support Services Market - Procurement Intelligence Report

Global Site Networks And Healthcare Systems Market - Procurement Intelligence Report

Global Clinical Trial Drug Supplies Market - Procurement Intelligence Report

Global Electronic Patient-Reported Outcome (ePRO) Services Market - Procurement Intelligence Report
Access this report and our entire procurement platform | Plans starting from USD 3000/ Year Buy Now
Copyright © 2026 Infiniti Research Limited. All Rights Reserved. Privacy Notice – Terms of Use – Sales and Subscription
Cookie Policy
The Site uses cookies to record users' preferences in relation to the functionality of accessibility. We, our Affiliates, and our Vendors may store and access cookies on a device, and process personal data including unique identifiers sent by a device, to personalise content, tailor, and report on advertising and to analyse our traffic. By clicking “I’m fine with this”, you are allowing the use of these cookies. You may change your settings based on a legitimate interest at any time, by selecting “Manage Settings” on our site. Please refer to the help guide of your browser for further information on cookies, including how to disable them. Review our Privacy & Cookie Notice.